|
Connect.
|
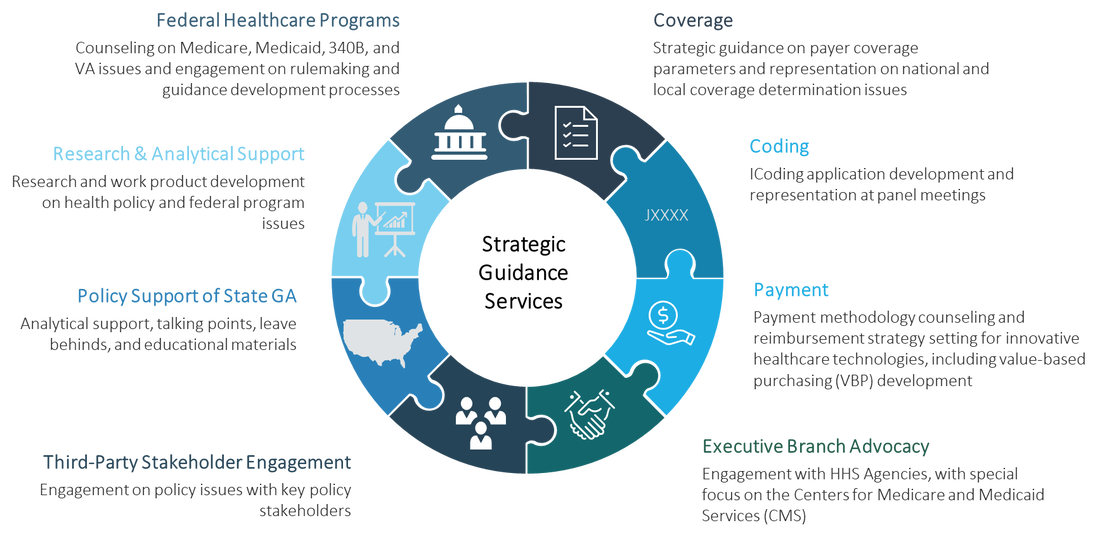
Our Health and Reimbursement Policy Group advisors possess deep yet broad health policy experience, working across a wide array of healthcare technology companies. They regularly provide strategic advice and tactical support for cutting-edge healthcare companies of all sizes. Their work spans both traditional health policy and coverage, coding, and payment issues. Our advisors also regularly represent clients before the Administration and other health policy stakeholders.
|
|
Health Policy Strategic Advising
|
As a Washington DC-based health policy firm, our advisors are at the forefront of emerging health policy developments and keep a strong pulse on shifts occurring in the healthcare landscape. With extensive experience in understanding how policy processes work in Washington and within the Administration, we are well-versed in assisting companies in navigating the complexities of health policy issues in federal healthcare programs and engaging on priority policy proposals for clients.
|
Our health policy strategic advising services are unique to each client and their technologies, as well as their long-term policy priorities. Our goal in strategic advising is to ensure that companies’ health policy strategic plans are attuned to the healthcare landscape, well-informed by existing regulations and guidance, and continually updated as developments in the landscape occur. These services typically are provided on an ongoing basis, with our team providing regular advice and support based on established priorities for the client.
|
Federal Healthcare Programs
|
K&A’s policy advisors have spent their careers immersed in policy aspects of the federal healthcare programs, particularly Medicare, Medicaid, 340B and TRICARE. We have deep understanding of the Medicare payment systems, such as the Medicare Physician Fee Schedule, the Medicare Inpatient Prospective Payment System, the ESRD Prospective Payment System, the Hospital Outpatient Prospective Payment System, and the Medicare Part D Program; the Medicaid Drug Rebate Program; the 340B Drug Pricing Program; and the TRICARE prescription drug program. We also remain at the forefront of value-based payment demonstrations and other changes in federal payment systems, such as the Quality Payment Program.
|
We assess and advise on health policy proposals in rulemakings and guidance relating to these programs and the impacts on our clients. Our advisors guide clients seeking to advance policy proposals under these systems, as well as those seeking to meet with the federal government. We also regularly conduct research and develop work product, such as presentations, to respond to client questions on these programs.
|
Coverage, Coding, and Payment
Our advisors provide support to clients on an array of coverage, coding, and payment strategy issues for healthcare technology companies:
|
Education
We regularly conduct educational presentations on the foundations of coverage, coding, and payment for healthcare technologies to assist companies in building tactical plans.
Our advisors are knowledgeable about the coverage standards that apply for healthcare technologies in federal healthcare programs and conduct assessments to identify threats and opportunities for pre-commercial technologies. We are experienced in working with the Centers for Medicare and Medicaid Services (CMS) on National Coverage Determination (NCD) policies, including representing clients through the National Coverage Analysis (NCA) process. Our advisors also assist clients in remaining abreast of changes occurring through the Local Coverage Determination (LCD) process with the Medicare Administrative Contractors (MACs). |
Coding
Our advisors support clients on a variety of coding applications and related services, such as:
|
Payment
Our advisors are well-versed in payment methodologies that apply to healthcare technologies across federal healthcare programs. We assist companies in understanding the cascade of payment methodologies that apply for newly commercialized technologies in federal healthcare programs. Our Group regularly assists clients in navigating Medicare’s complex payment systems, such as the Hospital Outpatient Prospective Payment System and the Medicare Physician Fee Schedule.
|
|
Gene & Cell Therapy
Our firm possess experience in representing cell and gene therapy companies on an array of health policy and coding, coverage, and payment matters. Our advisors are knowledgeable on the critical policy issues and developments for these therapies, such as genetic screening considerations, innovative payment priorities, existing system barriers, and care coordination complexities. We assist these companies in taking an agile approach to the shifting health policy landscape while advocating for policies and structural changes to better reflect the unique nature of these therapies.
|
|
Support for Pre-Commercial Companies
Our firm often works with pre-commercial companies several years in advance of their first launch, assisting companies in developing and executing on a strategic health policy plan. These services often involve the following:
|
|
Health Policy Presentations & Meeting Facilitation
Our advisors present on health policy matters at conferences, Board meetings, and client cross-functional team meetings. We also have experience facilitating cross-functional meetings to attain alignment on policy priorities and objectives, as well as serving as moderators at health policy-focused events.
|